
Share this post!
If I were a contestant on the Family Feud and 100 people were asked to name a nutrient-rich vegetable, I would slam that buzzer and offer up broccoli as the number one answer. Broccoli is a green, cruciferous vegetable in the Brassica family. Not only is it a great source of fiber, iron, potassium, calcium, selenium, magnesium, vitamins A, C, E, and K, several B vitamins, including folate, and even moderate amounts of protein, broccoli has an ace in the hole: sulforaphane.
Sulforaphane is a sulfur-containing phytonutrient that protects cells against oxidative damage and inflammation, and the best way to get it is by consuming broccoli and broccoli sprouts, yet no sulforaphane is present in these foods. Wait, what?
Yes, you read that right. Sulforaphane is a powerful nutrient we obtain from eating broccoli, though it doesn’t exist in raw, intact broccoli and broccoli sprouts. Read on to learn about sulforaphane’s transformation and health benefits.
Defining Terms
Before we get to sulforaphane, we need to understand the types of nutrients and enzymes that interact to form this magical nutrient. Let’s start with glucosinolates.
Glucosinolates
Glucosinolates are sulfur-containing compounds found in cruciferous vegetables and their seeds, such as broccoli, broccoli sprouts, Brussels sprouts, cabbage, horseradish, kale, and more. They are also found in the leaves, stems, and flowers of the Moringa plant.
Structurally, glucosinolates contain an amino acid derivative (often methionine), a glucose molecule, a sulfate group, and a varying carbon chain. A sulfur atom connects the glucose molecule to the rest of the structure. This is known as a “thio-linked glucose”.
These compounds are responsible for the pungent and bitter aromas and tastes of cruciferous vegetables. They are also an important part of the plant’s defense system. As the plant is damaged, such as by insects, chopping, chewing, or freezing and thawing, its cells break down allowing the glucosinolates to come into contact with an enzyme that catalyzes their transformation.
Myrosinase
Myrosinase is the enzyme that is present in plants with high glucosinolate content. It is a β-thioglucosidase enzyme, which is a fancy, scientific way to say that this enzyme breaks apart the thio-linked glucose in glucosinolates.
In the undisturbed plant, glucosinolates and myrosinase are kept in their own separate compartments. Damage to plant cells allows these components to mix and form a new category of compounds called isothiocyanates.
Sound familiar? That is because it is similar to the way allicin is formed when garlic cloves are similarly damaged.
An enzyme like myrosinase is a complex protein that is inactivated by heat. Therefore, cooking the vegetables prior to damaging the plant cells means that glucosinolates don’t get the chance to transform into isothiocyanates. However, this conversion also occurs in the human digestive tract, thanks to bacteria that produce the enzyme, myrosinase.
Isothiocyanates
Isothiocyanates are a class of compounds that contain this functional group: R-N=C=S, where R may be any form of carbon-based side chains, N is nitrogen, C is carbon, and S is sulfur. These molecules aren’t particularly stable in the body and they transform in the liver pretty rapidly.
Note in the following diagram the glucose molecule linked to the sulfur atom in the precursor molecule, whose removal is catalyzed by myrosinase.
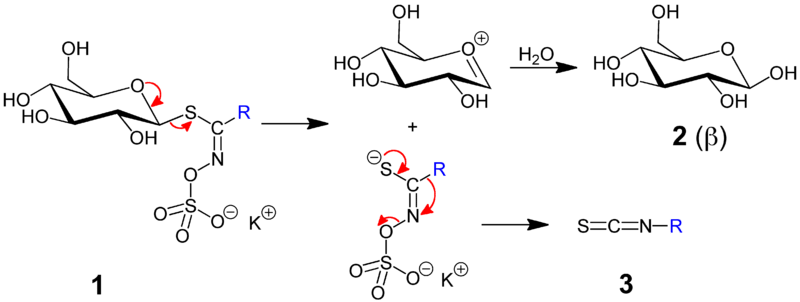
During their short-lived existence in the human body, isothiocyanates may cause several health benefits, including increased antioxidant activity, anti-inflammatory response, and activation of detoxification enzymes in the liver. They may even protect against cancer growth.
Each individual glucosinolate will form a unique isothiocyanate when exposed to myrosinase, such as the predominant glucosinolate in broccoli.
How Sulforaphane Forms in Broccoli
So now we have a formula:
Glucosinolates + Myrosinase → Isothiocyanates
Glucoraphanin is a glucosinolate present in high concentrations in broccoli and broccoli sprouts. Chopping, chewing, or freezing then thawing broccoli breaks down the plant cell walls and allows myrosinase to transform glucoraphanin to sulforaphane, an isothiocyanate.
Remember that myrosinase is heat sensitive, so cooking broccoli without disturbing the plant first will yield no sulforaphane, though myrosinase-producing gut bacteria may facilitate some conversion in the digestive tract.
Glucoraphanin and sulforaphane are both stable in the presence of heat, so there are ways to circumvent the loss of myrosinase and still reap the benefit of sulforaphane. For example, mustard seeds also contain myrosinase. Adding mustard or mustard powder to cooked broccoli reintroduces some myrosinase, which can mix with the glucoraphanin still present in cooked broccoli to form sulforaphane.
How Much Glucoraphanin is in Broccoli Products
The amount of sulforaphane capable of being produced from broccoli depends on the amount of glucoraphanin the seeds started with and the stage of growth the plant is in. This is highly variable based on the plant’s genetics and the environment in which the plant is grown. Broccoli seeds contain the highest amount of glucoraphanin, followed by the young sprouts, while mature broccoli contains the least amount.
It is difficult to standardize the amount of glucoraphanin present in food products, mainly because testing for this sort of thing is not typically done. However, supplements may be standardized for glucoraphanin content, sulforaphane content, or formulated as glucoraphanin plus myrosinase to enable the conversion.
Bioavailability of isothiocyanates, including sulforaphane, is greater than the glucosinolate precursor due to their fat solubility. It is evident that the presence of myrosinase greatly increases the availability of sulforaphane, whether from plant disruption, microbial contribution, or inclusion in supplemental formulas.
What are the Benefits of Sulforaphane?
Positive results from numerous clinical studies using sulforaphane have elevated this plant-based compound to a promising nutraceutical, which is a product derived from food sources that may have extra health benefits beyond the nutritional value of the food. Research shows the potential of sulforaphane to prevent and treat numerous conditions, including cancer, diabetes, skin disorders, cardiovascular conditions, autism, and aging. In addition, the growing popularity of nutraceuticals means that plant-based compounds like sulforaphane will remain a popular research topic.
Despite the variety of health conditions that are tested in research models, sulforaphane’s mechanisms of action target factors that are relevant for anyone, regardless of health status. These relevant factors are oxidative stress and inflammation, which cannot be avoided, but there are ways to improve the body’s ability to manage them. Here’s how sulforaphane may help.
Inducing the NRF2 Pathway
Although sulforaphane is not an antioxidant itself (at least not directly), one of the ways it benefits human health is by upregulating the body’s innate ability to handle oxidative stress. A major pathway the body uses to accomplish this is the Nrf2 pathway.
The Nrf2 pathway (nuclear factor erythroid 2-related factor-2) is very complicated and initiates the formation of numerous gene products (i.e. proteins and enzymes) related to the protection of the cell. In a nutshell, sulforaphane induces the Nrf2 pathway, which stimulates certain genes to produce proteins and enzymes that increase glutathione production (the body’s main innate antioxidant) and decrease inflammation.
Therefore, via a mechanism similar to allicin in garlic, sulforaphane induces a strong protective effect in cells by creating a small stress that results in the cell unleashing a powerful antioxidant and anti-inflammatory response. By minimizing the accumulation of oxidative stress and inflammation, sulforaphane may help to prevent the onset of pathological conditions, such as cancer, heart disease, and cognitive dysfunction.
Other Actions of Sulforaphane Include:
- Accelerating detoxification and excretion of potential carcinogens and pollutants
- Increasing glutathione levels in the brain
- Reducing LDL cholesterol
- Decreasing fasting glucose levels
Is Sulforaphane Safe?
Luckily, humans have an innate aversion to bitter foods as a protective measure against toxic compounds. Therefore, it is unlikely that people will consume broccoli or broccoli sprouts to the point of toxicity for sulforaphane. However, with supplements becoming increasingly available it is worthy of caution.
Research into sulforaphane and its effects is still relatively new, but some studies in mice are showing adverse effects at doses above 150 mg/kg, including sedation, impaired motor function, hypothermia, or death. In addition, some cancer studies in mice show increased tumor development when sulforaphane was administered after cancer induction, but had protective effects when administered before, suggesting a more preventative role.
Another concern of isothiocyanate consumption is the potential interference of these compounds with the production of thyroid hormone, particularly in those with hypothyroidism and/or an iodine deficiency, as isothiocyanates may compete with the iodine transporter in the thyroid gland. Fortunately, many research studies, including this one, show that broccoli products are safe for those with thyroid conditions and may even improve its function by increasing antioxidant capacity. If isothiocyanates are a concern, avoiding excessive consumption of cruciferous vegetables and/or cooking them to inactivate myrosinase may be prudent.
Finally, broccoli sprouts (the highest food-based source of sulforaphane) may not be advisable for children, the elderly, or those with compromised immune systems due to the risk of bacterial contamination from improper sanitation or storage.
Other Sources of Sulforaphane?
Not all cruciferous vegetables contain glucoraphanin, or at levels similar to broccoli, therefore they won’t yield sulforaphane at all or they may produce it in much smaller amounts, though other glucosinolates may convey similar health benefits. Some cruciferous vegetables, such as Brussels sprouts and kale, may contain a small amount of glucoraphanin, but the main source from food is broccoli.

Ideas for Food-Based Sulforaphane Intake
The richest source of glucoraphanin comes from young broccoli sprouts, which may result in 10-100 times the sulforaphane activity as mature broccoli. They can be found at some grocery stores, or even made at home with simple tools and a few minutes per day. If you want to try sprouting at home, I highly recommend Dr. Rhonda Patrick’s Sprouting Guide, which you can get by going to her website here and signing up for her newsletter. You can add them to salads, soups, smoothies, and more.
Here’s a recipe that incorporates raw broccoli, which preserves the myrosinase. Plus, it has a mustard based dressing for a little extra enzyme oomph!
Honey Mustard Broccoli Salad
The following recipe is adapted from Cookie and Kate’s Favorite Broccoli Salad.
Ingredients:
- 1 pound broccoli, finely chopped
- ½ cup red onion, finely chopped
- Optional additions: ½ cup sunflower seeds, ½ cup raisins, ½ cup chopped bacon, ½ cup cheddar cheese
Dressing:
- ⅓ cup extra virgin olive oil
- 2 T apple cider vinegar
- 2 T dijon or brown mustard
- 1 T honey (or maple syrup)
- 1 clove garlic, minced
- salt to taste
Directions: Combine salad ingredients of choice in a large bowl. Whisk together dressing ingredients in a small bowl then mix with the salad. This dish is best if allowed to marinate for an hour in the fridge or overnight prior to serving.
Enjoy and chew well to maximize sulforaphane intake!
About the Author:
Karyn Lane is a current student of NTI’s Nutrition Therapist Master Program. She finds her chemistry degree a useful tool in her study of nutrition and loves to treat herself as a laboratory for new recipes and cooking techniques. You can follow her on Instagram @feel.alive.nourishment.
Image:
Image by Engin_Akyurt is free for use by Pixabay
Image by Jü is licensed under CC BY-SA 3.0
Image by jack3928 is free for use by Pixabay
Learn more about our school by attending an informational webinar.
Share this post!




















