
Share this post!
A Course in Nutrition: Learning Your A, B, C’s
Welcome back to part 10 of our multi-part series on vitamins.
In the last blog of the series, we talked about vitamin D.
We highlighted:
- Forms of vitamin D and their activation
- How it regulates calcium metabolism and supports healthy bones
- Its roles in immune function
- How deficiency may affect our health
- Recommended intake and optimal levels
- Sunlight exposure and food sources, plus a customizable salad recipe
Next, we move on to another fat-soluble vitamin: Vitamin E.
Vitamin E: An Assortment of Related Compounds
Rather than being one isolated chemical structure, vitamin E refers to a collection of similar compounds called tocopherols and tocotrienols. There are 4 isomers within each of these subgroups – alpha, beta, gamma, and delta – making 8 specific molecules that fall under the vitamin E heading.
The following image depicts a tocotrienol. Note the double bonds in red. If no double bonds are present on the right side of the structure (i.e. take out the red lines), this structure represents a tocopherol. The most well-known and well-studied version of vitamin E is alpha-tocopherol, in which all of the R groups are methyl (-CH3) groups. In the beta, gamma, and delta versions of tocopherols or tocotrienols, these groups are either methyl groups or hydrogen atoms, making them very similar structurally.
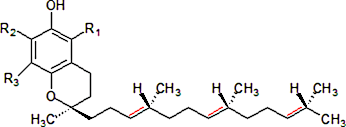
The hydroxyl (-OH) containing aromatic ring structure on the left of this molecule, called a chromanol, make this side of the molecule soluble in water, while the side chain on the right is soluble in fat. Therefore, this amphipathic characteristic makes the molecule uniquely suited to interact with cell membranes, as the outer layer of the cell is made of phospholipids with similar properties.
As you can see, there’s a lot going on structurally in vitamin E that can produce a lot of natural variation in the molecular forms. Though interesting to me as someone who enjoys organic chemistry (and hopefully you do too), they’re not critical to know in assessing the nutritional function of vitamin E. There is one structural concept, however, that is important when determining the biological activity of natural versus synthetic vitamin E.
Natural vs Synthetic Vitamin E
Tocopherols contain 3 chiral carbons, which means that the carbon is attached to 4 different substituents. These substituents, which are ranked according to priority, can be oriented in different ways in space, much like the way your fingers are attached on your right versus left hand. If the substituents rotate to the right according to their priority, the molecule is notated with a “d” for dextrorotatory. If the substituents rotate to the left, it is notated with an “l” for levorotatory.
In nature, the substituents of alpha-tocopherol’s chiral carbons always rotate to the right, making it d-alpha-tocopherol. Synthetic alpha-tocopherol contains mixtures of d and l isomers in equal proportions, which is called dl-alpha-tocopherol. D-alpha-tocopherol is the most biologically active form, therefore you would need twice the amount of synthetic vitamin E to achieve the same activity as natural vitamin E.
Alpha-tocopherol is the main form associated with benefits to human health and it is often used synonymously with vitamin E. Tocotrienols have varying levels of biological activity, yet they have not been as widely studied, most likely because of their lower bioavailability.
Vitamin E is an Antioxidant
This vitamin neutralizes free radicals directly by donating an electron. Some notable recipients of vitamin E’s generous donation are other antioxidants themselves, particularly vitamin C, glutathione, and coenzyme Q10. These molecules have each other’s back – when one becomes oxidized, the others swoop in to donate an electron to restore them to their reduced state so that they may continue the fight against oxidative damage.
Vitamin E protects the phospholipids of cell membranes thanks to its solubility in fat, though these aren’t the only lipid-based entities that vitamin E protects. Lipoproteins, such as VLDL, LDL, and HDL particles, are special vehicles consisting of proteins and lipid molecules whose job is to transport fat and cholesterol throughout the body via the water-based medium of blood. Vitamin E protects these particles from oxidation, which makes them sticky and prone to clumping.
Outside of the body, vitamin E protects the fats in our food as well, which is especially useful for polyunsaturated fats like omega-3’s whose double bonds are especially vulnerable to oxidation. Not to mention its original role as protection for plants.
Vitamin E and Health Conditions
Research regarding vitamin E’s support to the health of the body are mainly focused on the following conditions:
- Cardiovascular disease
- Cancer
- Eye conditions, such as age-related macular degeneration
- Cognitive decline
It’s worth noting that many of the studies related to these conditions show mixed results of E’s effectiveness and even evidence of some harm associated with vitamin E supplementation in some cases. There are many variables that go into research of this kind, particularly the use of isolated alpha-tocopherol versus the full mixture of vitamin E forms.
In addition, vitamin E supports a healthy immune system, not only for its ability to reduce oxidative stress, but also because it may play a role in cell signaling, improving immune cell function, and regulating gene expression.
Vitamin E Deficiency
According to recent National Health and Nutrition Examination Surveys (NHANES), intakes of vitamin E are low for the majority of the population – as many as 84% of us! Typical American diets are lacking in nuts, seeds, and green leafy vegetables, which you’ll see shortly are main sources of this nutrient.
Though averages intakes are notoriously low, overt deficiency symptoms are rare. These include:
- Neuropathy and nerve damage
- Degenerative diseases of the nervous system
- Loss of vision
- Immune system impairment
People with congenital conditions that impair the absorption of fat in the digestive tract are at greatest risk for vitamin E deficiency symptoms. Those with gall bladder or liver issues may also have difficulty absorbing enough vitamin E from the diet. Water-soluble forms, such as tocopheryl polyethylene glycol succinate, are available for those with serious fat absorption issues.
Recommended Intakes
The recommended dietary allowance (RDA) for vitamin E is 15 mg/day for adults, though some suggest that higher intakes would be beneficial for the elderly. Often you will find vitamin E listed as alpha-tocopherol equivalents (ATEs) on dietary labels if they are listed.
Recently, the FDA changed the labeling requirements for vitamin E from international units (IUs) to milligrams. Here’s how to convert IUs to mg.
- Natural vitamin E: 1 IU = 0.67 mg ATE or 1 mg ATE = 1.49 IU
- Synthetic vitamin E: 1 IU = 0.45 mg ATE or 1 mg ATE = 2.22 IU
The tolerable upper limit (UL) is set at 1000 mg/day based on toxicity risks including excessive bleeding or inability to form clots properly. Adverse effects come from supplemental doses, not from vitamin E in food.
Interestingly, the form of vitamin E that is most prevalent in American diets is gamma-tocopherol, not alpha-tocopherol, which is used to quantify vitamin E intake. Let’s take a closer look at food sources of vitamin E.

Food Sources of Vitamin E
Plants are the only species capable of synthesizing vitamin E, so the predominant food sources of this nutrient are plant-based. Animal-based foods with vitamin E are a result of the animal eating plants, or eating other animals that consumed plants.
Plant-based sources of vitamin E include:
- Sunflower Seeds
- Almonds
- Hazelnuts
- Peanuts/peanut butter
- Dark leafy greens – spinach, kale, turnip greens, mustard greens, chard
- Avocado
- Olives/olive oil
- Chili peppers
- Broccoli
- Asparagus
- Kiwi
- Mango
- Tomato
Gamma-tocopherol is abundant in the seeds of plants, including corn, soybean, and sesame seeds, and oils or other products made from them. Walnuts, pecans, and peanuts are also rich sources of gamma-tocopherol.
Animal-based sources of vitamin E include:
- Shrimp
- Abalone (snails)
- Goose meat
- Atlantic salmon
- Rainbow trout
- Egg yolk

Supplementation
Supplemental forms of vitamin E are most commonly alpha-tocopherol alone, either in natural or synthetic form, though some contain mixed tocopherols and tocotrienols. Most supplemental forms provide doses in excess of the RDA.
Often vitamin E in supplemental form is bound with another molecule to preserve its shelf-life in a process called chelation. Chelates of vitamin E are typically acetic acid (alpha-tocopheryl acetate) or succinic acid (alpha-tocopheryl succinate). Humans can break these bonds in the digestive tract and absorb alpha-tocopherol as efficiently as unbound forms.
Due to the blood-thinning properties of vitamin E, the use of supplementation is contraindicated with medications such as anticoagulants, antiplatelet drugs, and some cholesterol-lowering medications. Vitamin E is also not recommended during chemotherapy treatment, as it may reduce the effectiveness of these drugs.
Savory Almond Spread
Here’s a recipe for a nut-based dip adapted from Julie Piatt’s This Cheese is Nuts cookbook. One serving (about 1/4th of the recipe) contains 8 mg of vitamin E, about 40% of the RDA.
Ingredients:
- 1 cup soaked almonds
- 2 tbsp nutritional yeast
- 1 tbsp chili garlic sauce
- 1 tsp garlic powder
- 1 tsp salt
Directions:
Soak the almonds overnight in the refrigerator. Drain almonds and combine them in a food processor with the remaining ingredients. Add small amounts of water if necessary to achieve desired consistency.
Enjoy with crackers, on avocado toast (for extra vitamin E!), as a salad topping, or with sliced veggies.
Nuts aren’t your thing? Try this vitamin E-rich sunflower seed spread from NTI’s recipe archives. Happy snacking!
General reference: National Institutes of Health – Office of Dietary Supplements
About the author:
Karyn Lane is a current student of NTI’s Nutrition Therapist Master Program. She finds her chemistry degree a useful tool in her study of nutrition and loves to treat herself as a laboratory for new recipes and cooking techniques. You can follow her on Instagram @feel.alive.nourishment.
Image:
Image by Kathas_Fotos is free for use by Pixabay
Image by Yohan is licensed under CC BY-SA 3.0
Image by congerdesign is free for use by Pixabay
Image by buntysmum is free for use by Pixabay
Learn more about our school by attending an informational webinar.
Share this post!




















